Proofreading experimentally assigned stereochemistry through Q2MM predictions in Pd-catalyzed allylic aminations | Nature Communications

Palladium-Catalyzed Decarboxylative Asymmetric Allylic Alkylation of Enol Carbonates | Journal of the American Chemical Society

Exploiting the Versatility of Palladium Catalysis: A Modern Toolbox for Cascade Reactions - Mondal - 2021 - European Journal of Organic Chemistry - Wiley Online Library
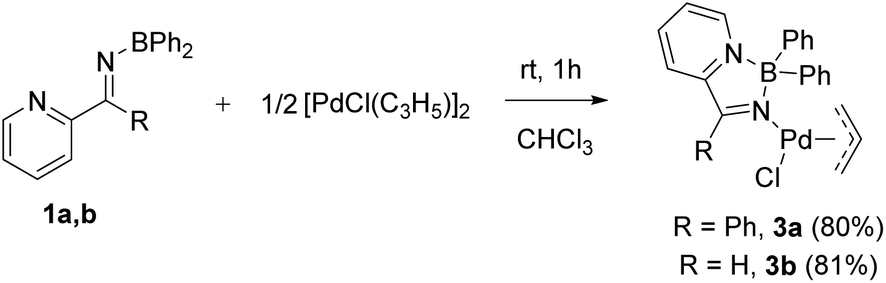
Synthesis, structure and palladium coordination of ambiphilic, pyridine- and phosphine-tethered N -boryl imine ligands - Dalton Transactions (RSC Publishing) DOI:10.1039/C8DT05100C

Palladium-catalyzed arylation of thiophene using PdCl(C 3 H 5 )(dppb)... | Download Scientific Diagram
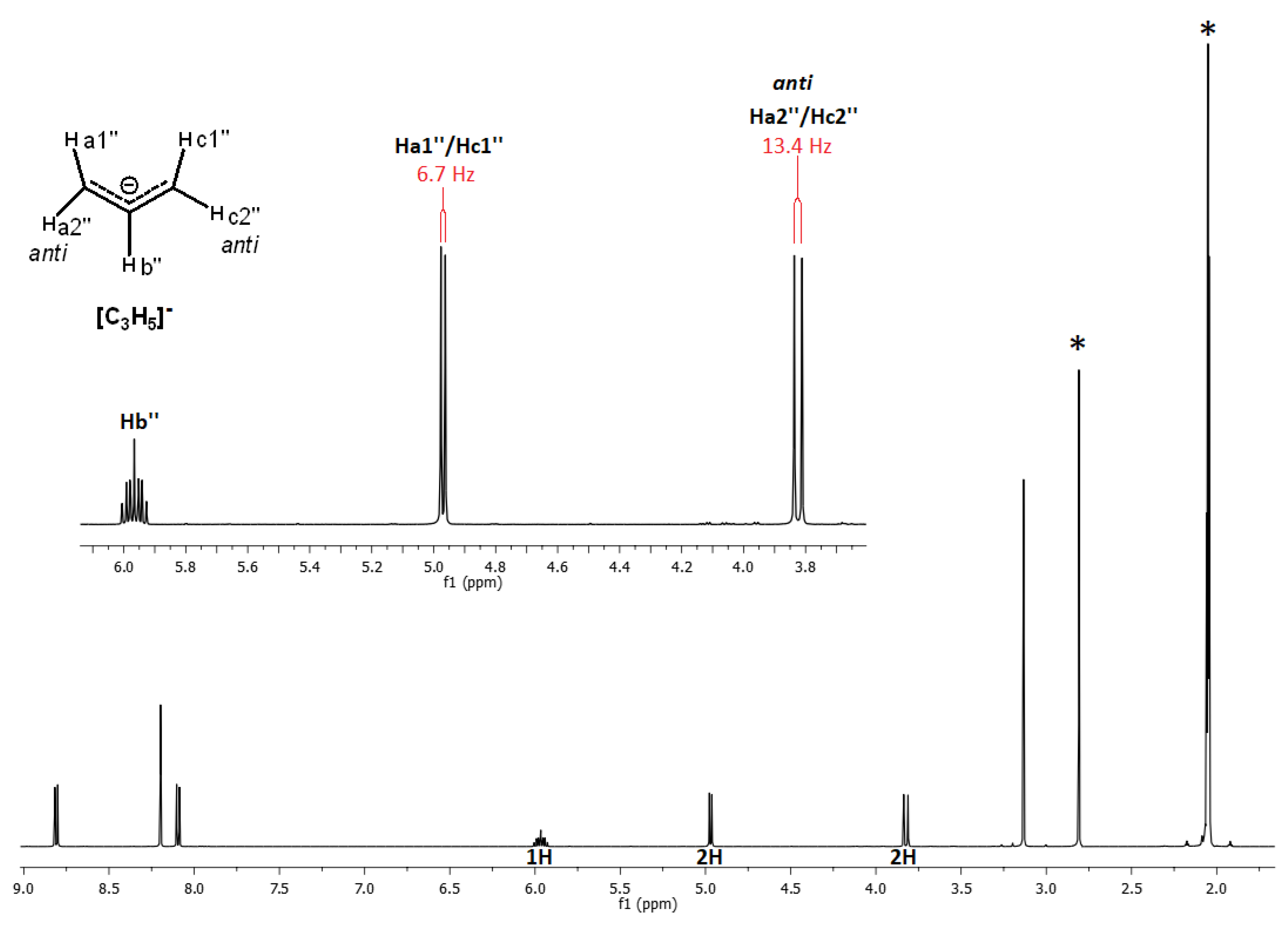
Chemistry | Free Full-Text | Synthesis, Characterization and Photophysical Properties of Mixed Ligand (η3-Allyl)palladium(II) Complexes with N,N’Aromatic Diimines
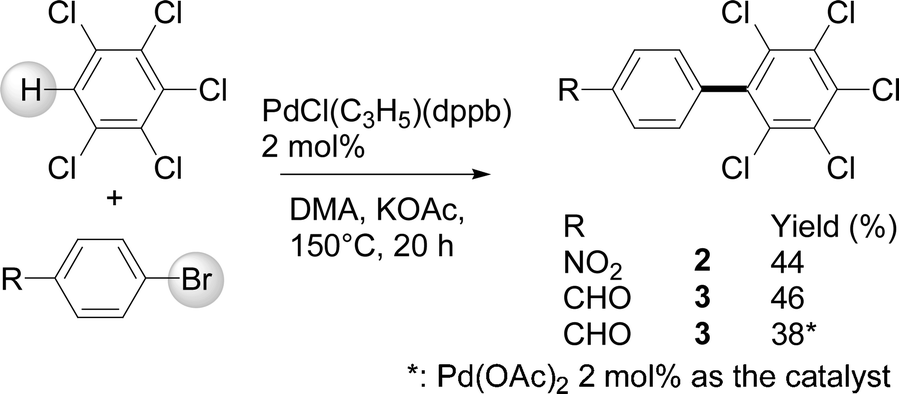
Reactivity of C–H bonds of polychlorobenzenes for palladium-catalysed direct arylations with aryl bromides - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C3CY00757J

Palladium‐Catalyzed Arylfluorination of Alkenes: A Powerful New Approach to Organofluorine Compounds - Remete - 2022 - Chemistry – A European Journal - Wiley Online Library
- - SpectraBase Palladium, [(1,2,3,4,5-.eta.)-1,2,3,4,5-pentamethyl-2,4-cyclopentadien-1-yl](.eta.3-2-propenyl)- - SpectraBase](https://spectrabase.com/api/compound/5FEwpoRDlM1.png?ph=true&h=300&w=382)
Palladium, [(1,2,3,4,5-.eta.)-1,2,3,4,5-pentamethyl-2,4-cyclopentadien-1-yl](.eta.3-2-propenyl)- - SpectraBase

Regio- and enantioselective umpolung gem-difluoroallylation of hydrazones via palladium catalysis enabled by N-heterocyclic carbene ligand | Nature Communications
![EurJOC on X: "Palladium-Catalyzed C-H Bond Functionalization of Benzo[1,2-b:4,5-b']dithiophene-4,8-dione: A One Step Access to 2-Arylbenzo[1,2-b:4,5-b']dithiophene-4,8-diones by Pierre Frere, Veronique Guerchais, Henri Doucet and co-workers #OpenAccess ... EurJOC on X: "Palladium-Catalyzed C-H Bond Functionalization of Benzo[1,2-b:4,5-b']dithiophene-4,8-dione: A One Step Access to 2-Arylbenzo[1,2-b:4,5-b']dithiophene-4,8-diones by Pierre Frere, Veronique Guerchais, Henri Doucet and co-workers #OpenAccess ...](https://pbs.twimg.com/media/FyFDq8iWAAAeC_c.png)
EurJOC on X: "Palladium-Catalyzed C-H Bond Functionalization of Benzo[1,2-b:4,5-b']dithiophene-4,8-dione: A One Step Access to 2-Arylbenzo[1,2-b:4,5-b']dithiophene-4,8-diones by Pierre Frere, Veronique Guerchais, Henri Doucet and co-workers #OpenAccess ...

Synthesis of 4‐Formyl‐2‐arylbenzofuran Derivatives by PdCl(C3H5)dppb‐Catalyzed Tandem Sonogashira Coupling‐Cyclization under Microwave Irradiation ‐ Application to the Synthesis of Viniferifuran Analogues - Vo - 2017 - ChemistrySelect - Wiley Online ...

Palladium-Catalyzed Desymmetrization of Silacyclobutanes with Alkynes: Enantioselective Synthesis of Silicon-Stereogenic 1-Sila-2-cyclohexenes and Mechanistic Considerations | Organic Letters

Palladium-catalyzed asymmetric tandem allylic substitution using chiral 2-(phosphinophenyl)pyridine ligand - ScienceDirect

Palladium-Catalyzed Trimethylenemethane Cycloaddition of Olefins Activated by the σ-Electron-Withdrawing Trifluoromethyl Group. | Semantic Scholar
![Recent Advances in Palladium-Catalyzed [4 + n] Cycloaddition of Lactones, Benzoxazinanones, Allylic Carbonates, and Vinyloxetanes | Topics in Current Chemistry Recent Advances in Palladium-Catalyzed [4 + n] Cycloaddition of Lactones, Benzoxazinanones, Allylic Carbonates, and Vinyloxetanes | Topics in Current Chemistry](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs41061-023-00442-9/MediaObjects/41061_2023_442_Sch52_HTML.png)
Recent Advances in Palladium-Catalyzed [4 + n] Cycloaddition of Lactones, Benzoxazinanones, Allylic Carbonates, and Vinyloxetanes | Topics in Current Chemistry


![photolysis o f [($-C3H5) (@-C5H5)Pd] on pol yimide substrates. | Download Scientific Diagram photolysis o f [($-C3H5) (@-C5H5)Pd] on pol yimide substrates. | Download Scientific Diagram](https://www.researchgate.net/profile/James-Spencer-3/publication/266349735/figure/fig3/AS:669279976452112@1536580189751/photolysis-o-f-C3H5-C5H5Pd-on-pol-yimide-substrates_Q320.jpg)