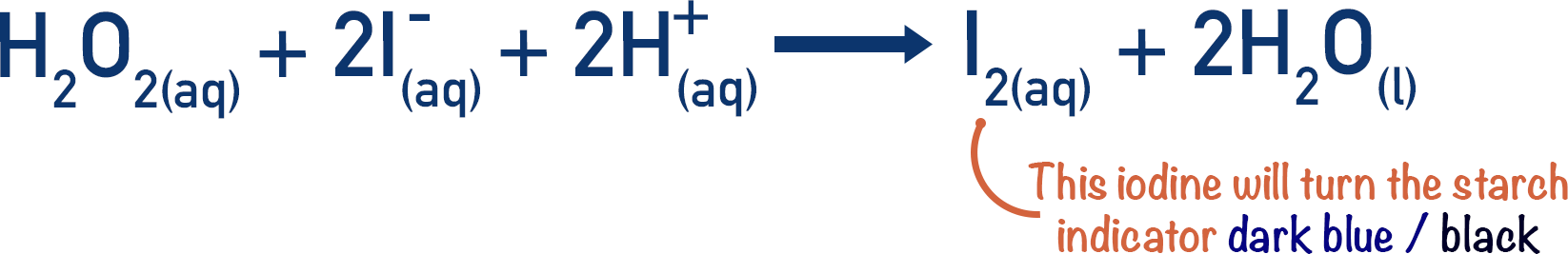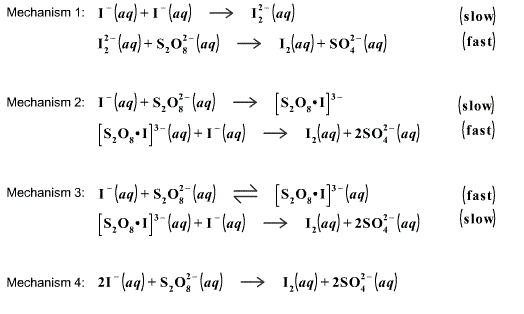SOLVED: Reaction Rates Concentrations: Effect of Reactant Concentrations Demo: "Iodine Clock" Reaction Iodate Variation IO3- + 3 HSO3- â†' 1-+ 3 SO42- + 3 H2O (slow) IO3- + 5I- + 6 H+

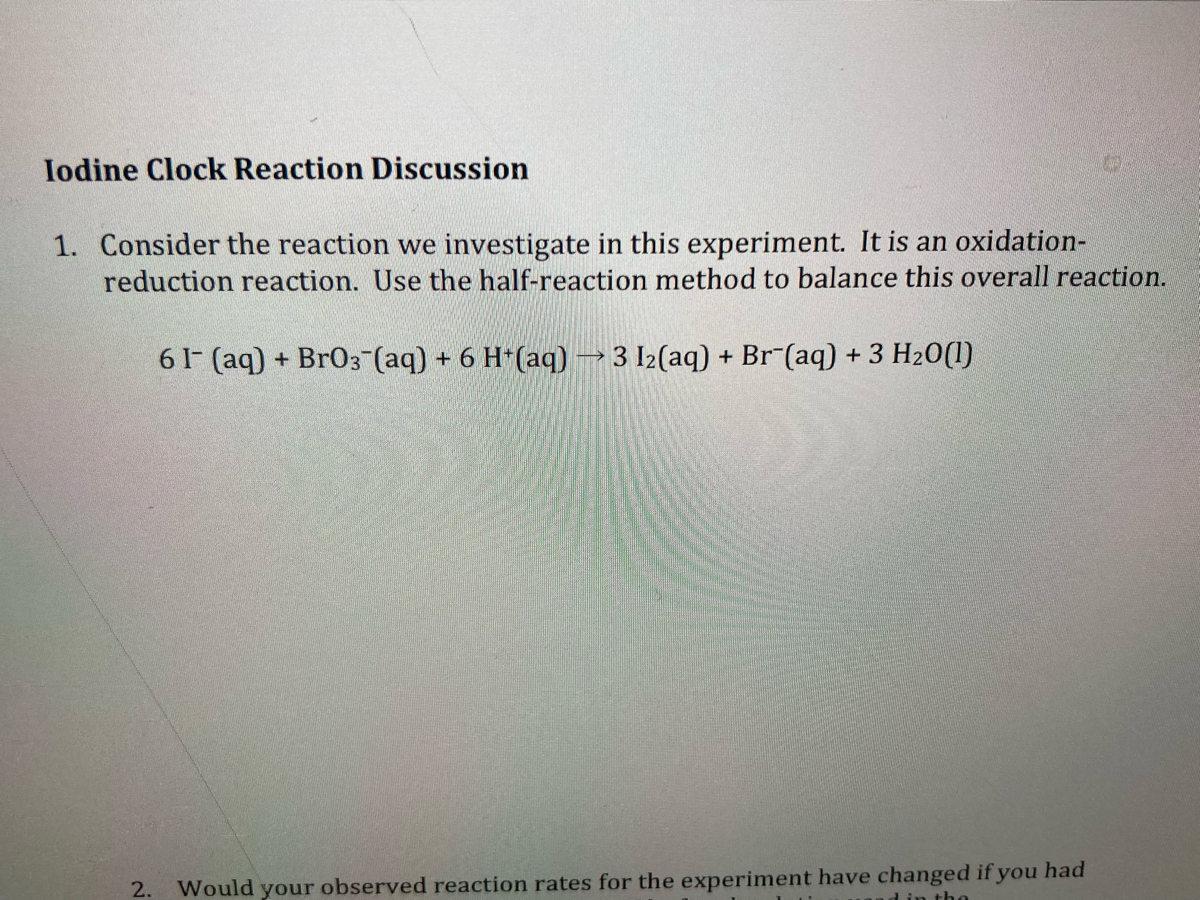
Exp16calcs W - lab report - Chemical Kinetics: The Iodine-Clock Reaction: S 2 O 82 −(aq) + 2 I−(aq) - Studocu

PPT - In the iodine clock reaction, there are really two processes happening simultaneously. The first is a slow reaction PowerPoint Presentation - ID:39733

A Closer Examination of the Mechanism of the Hydrogen Peroxide Iodine-Clock Reaction with Respect to the Role of Hypoiodite Species | Journal of Chemical Education
![SOLVED: The rate law for the iodine clock reaction is given by: Rate = k[IO3-] [I-]2[H+]^2. a) This reaction is third order with respect to H+. b) This reaction is first order SOLVED: The rate law for the iodine clock reaction is given by: Rate = k[IO3-] [I-]2[H+]^2. a) This reaction is third order with respect to H+. b) This reaction is first order](https://cdn.numerade.com/ask_previews/494c958b-2794-4a7f-861f-022f5f445939_large.jpg)
SOLVED: The rate law for the iodine clock reaction is given by: Rate = k[IO3-] [I-]2[H+]^2. a) This reaction is third order with respect to H+. b) This reaction is first order