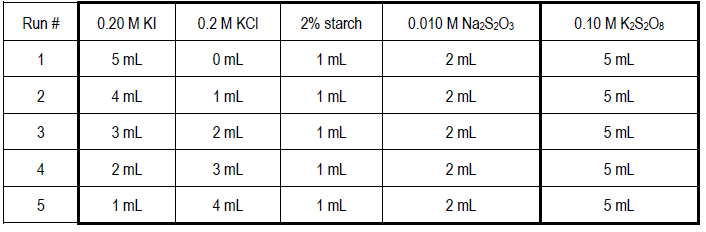
![A clock reaction is run at 20 �C with sev[{Image src='img20401705385059449876496.jpg' alt='' caption=''}]eral different mixtures of iodide, sodium bromate and acid, to form iodine. Thiosulfate is used | Homework.Study.com A clock reaction is run at 20 �C with sev[{Image src='img20401705385059449876496.jpg' alt='' caption=''}]eral different mixtures of iodide, sodium bromate and acid, to form iodine. Thiosulfate is used | Homework.Study.com](https://homework.study.com/cimages/multimages/16/img20401705385059449876496.jpg)
A clock reaction is run at 20 �C with sev[{Image src='img20401705385059449876496.jpg' alt='' caption=''}]eral different mixtures of iodide, sodium bromate and acid, to form iodine. Thiosulfate is used | Homework.Study.com

OneClass: In the Rate of an Iodine Clock Reaction experiment, the following data were collected by a ...

SOLVED: 3. Write the overall rate law for the iodine clock reaction below. What is the overall rate order of the main reaction? 5. Hydrogen gas reduces NO to N2 according to

SOLVED: Reaction Rates Concentrations: Effect of Reactant Concentrations Demo: "Iodine Clock" Reaction Iodate Variation IO3- + 3 HSO3- â†' 1-+ 3 SO42- + 3 H2O (slow) IO3- + 5I- + 6 H+

Autocatalysis-Driven Clock Reaction III: Clarifying the Kinetics and Mechanism of the Thiourea Dioxide–Iodate Reaction in an Acidic Medium | The Journal of Physical Chemistry A




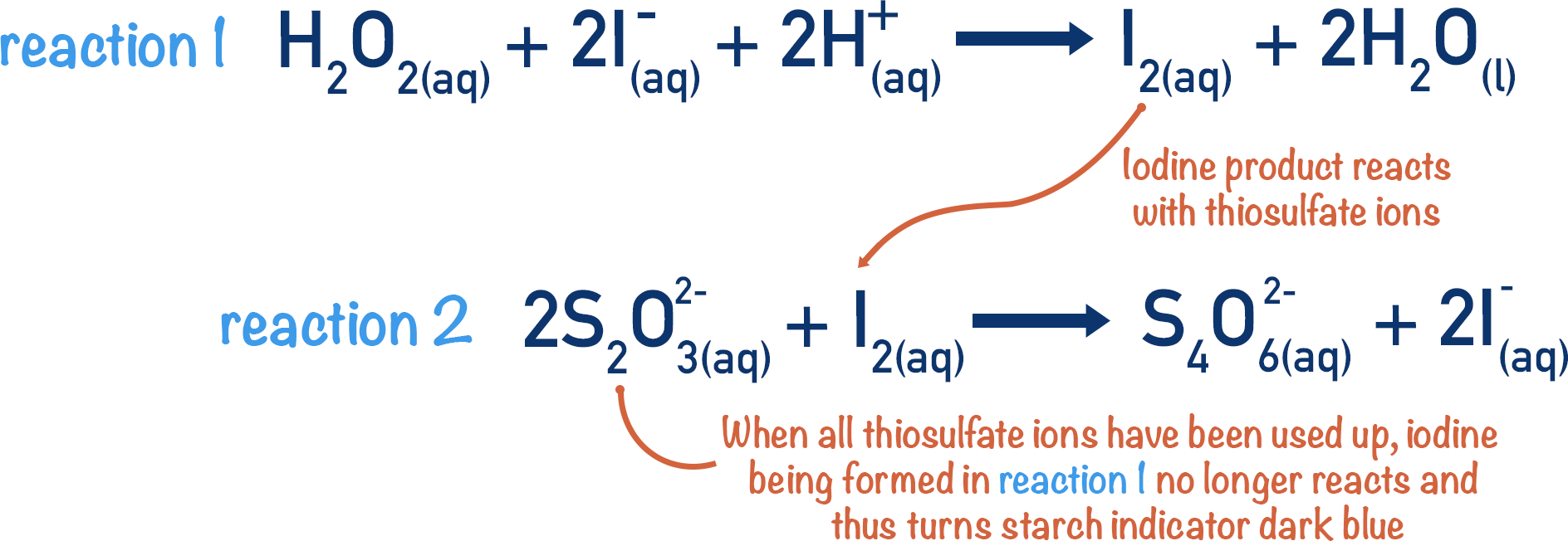

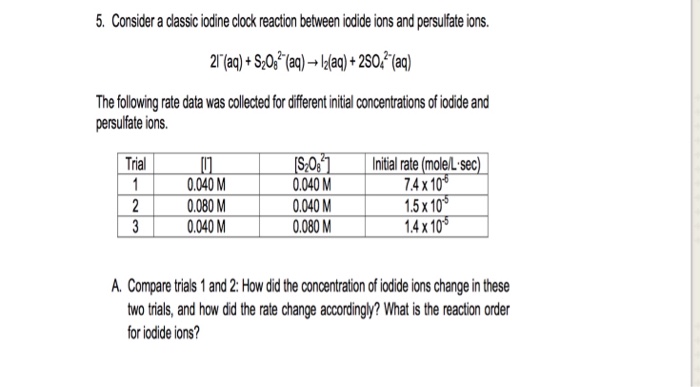
![Solved The iodine clock reaction Test [KIO3] (M) [NaHSO3] | Chegg.com Solved The iodine clock reaction Test [KIO3] (M) [NaHSO3] | Chegg.com](https://media.cheggcdn.com/media/4ea/4ea146b7-b7f4-4915-88de-6a4373a115e2/phpLZvxWR)