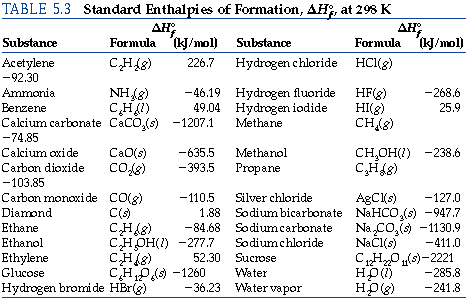
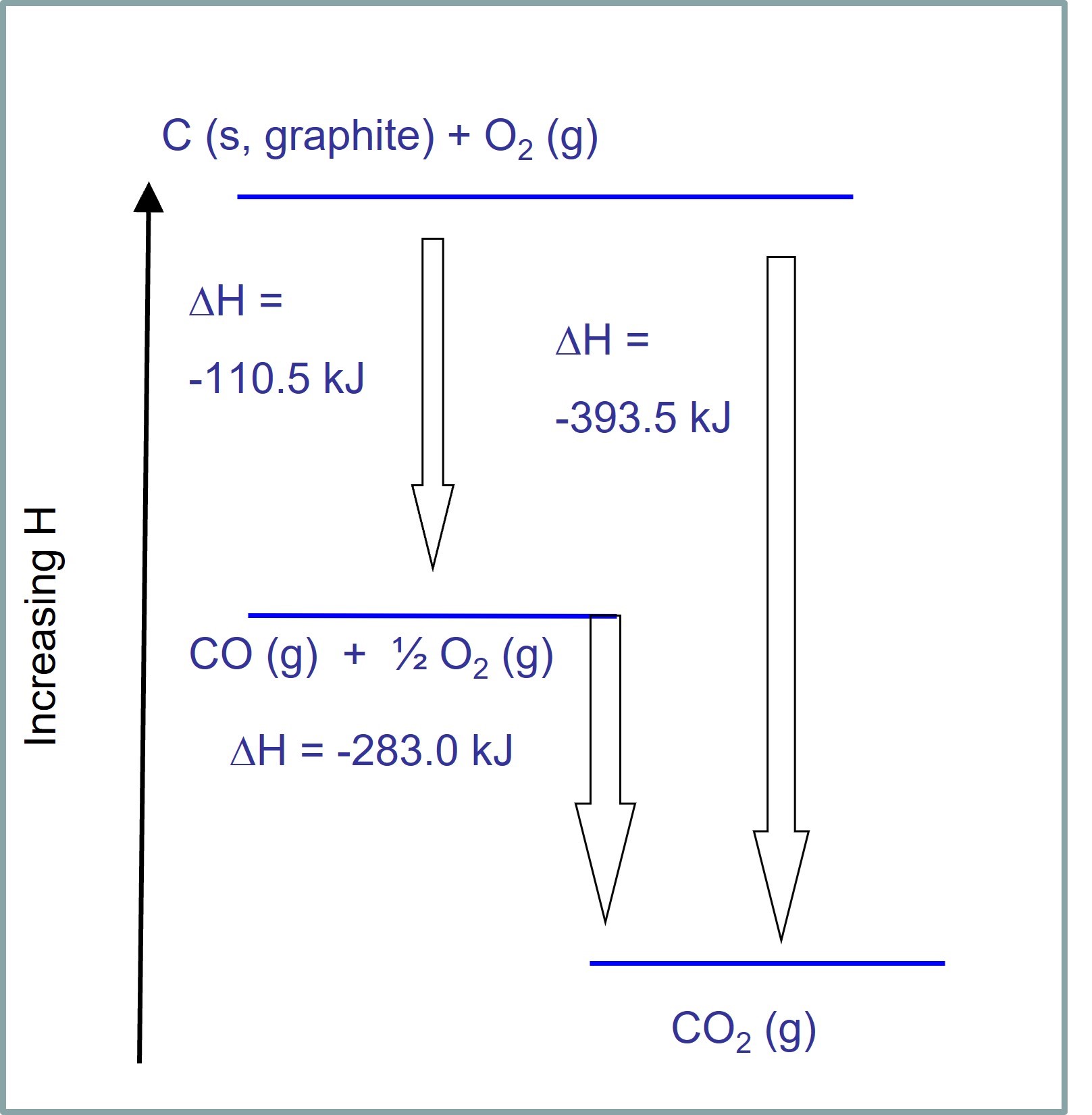
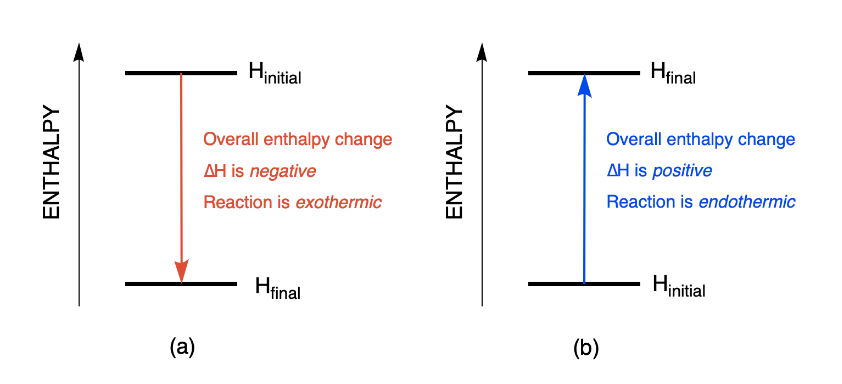
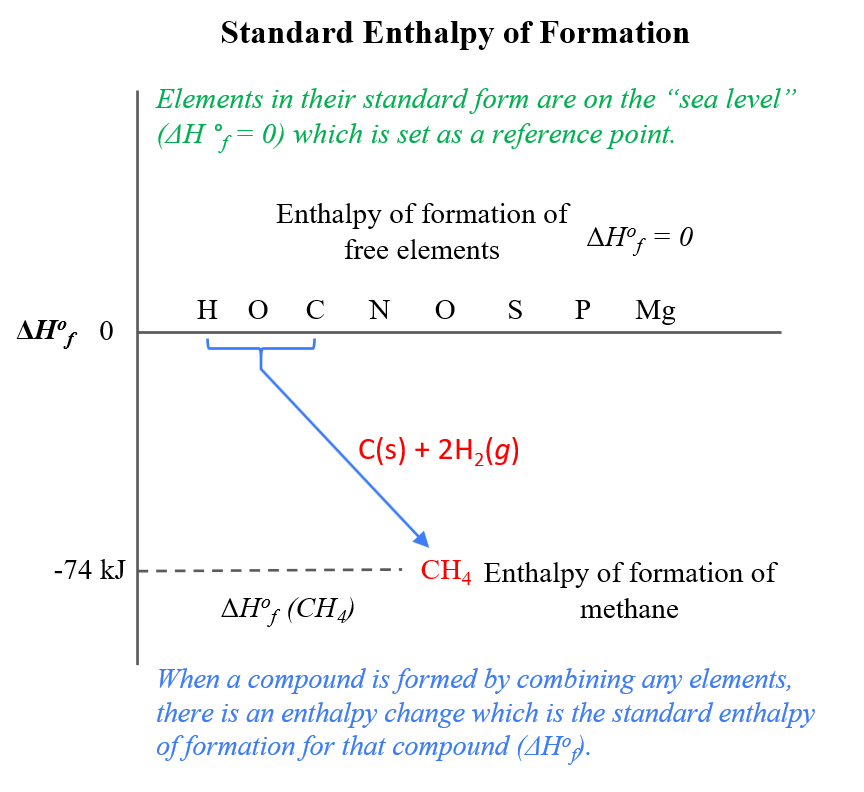
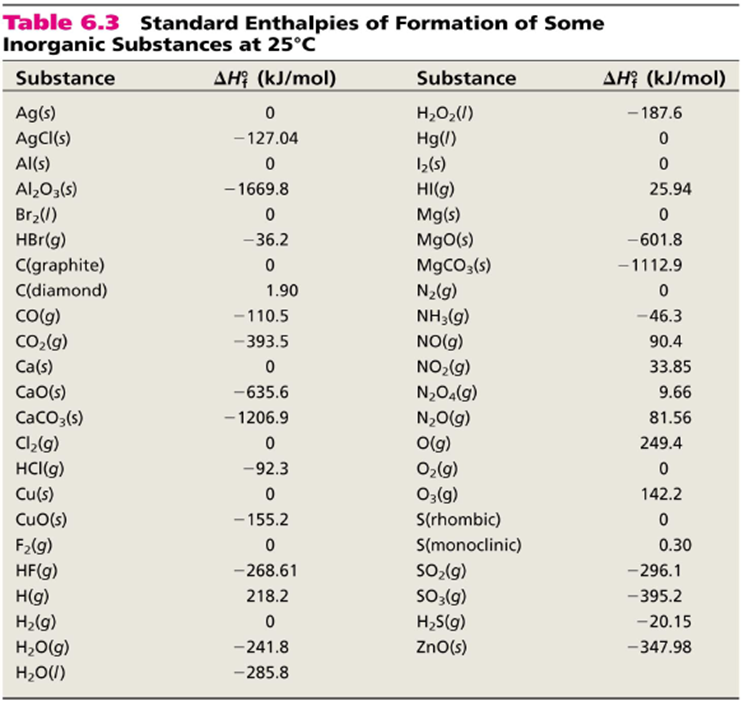
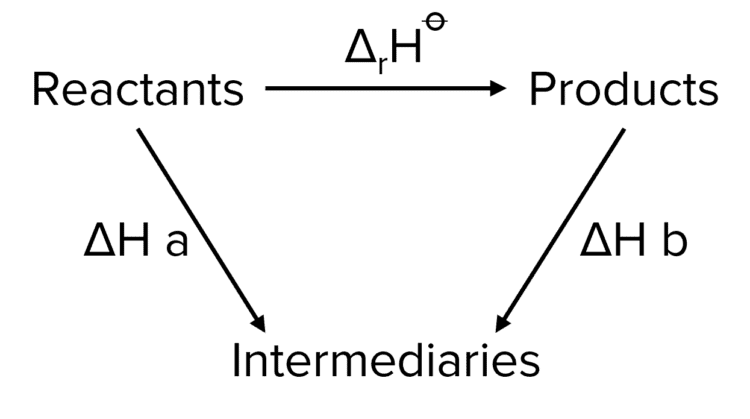
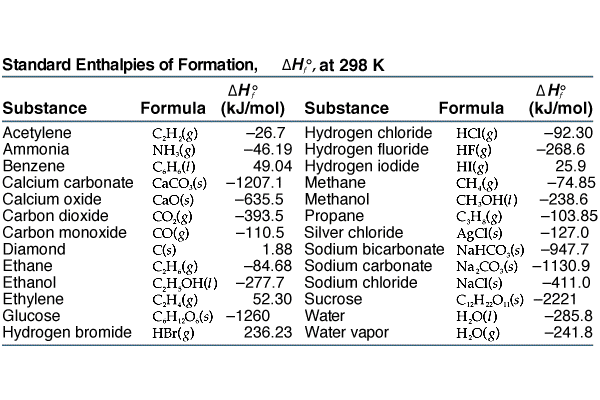
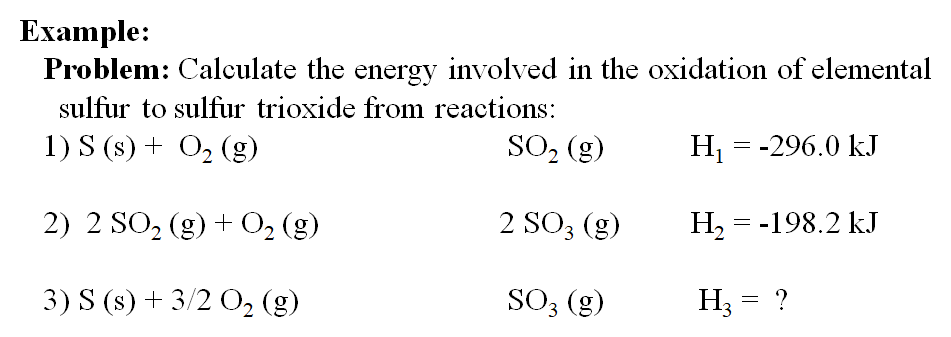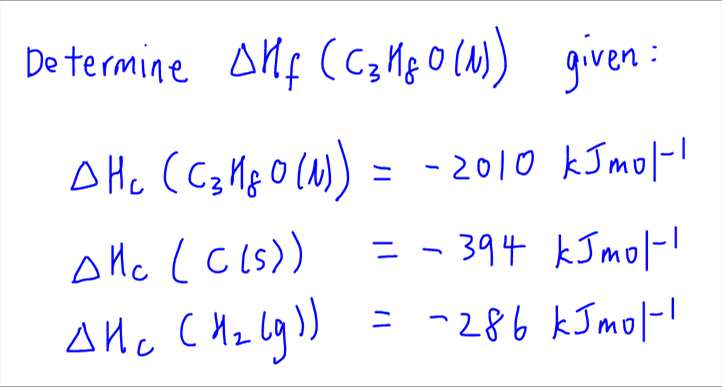The basis for calculating enthalpies of reaction is known as Hess's law: the overall enthalpy change in a reaction is equal to the sum of enthalpy changes. - ppt download

What is the overall enthalpy change DHrxn for the system? -1,300 kJ -300 kJ 300 kJ 1,300 kJ - brainly.com

Question Video: Determining a Standard Enthalpy Change Given the Standard Enthlapy of Fusion | Nagwa