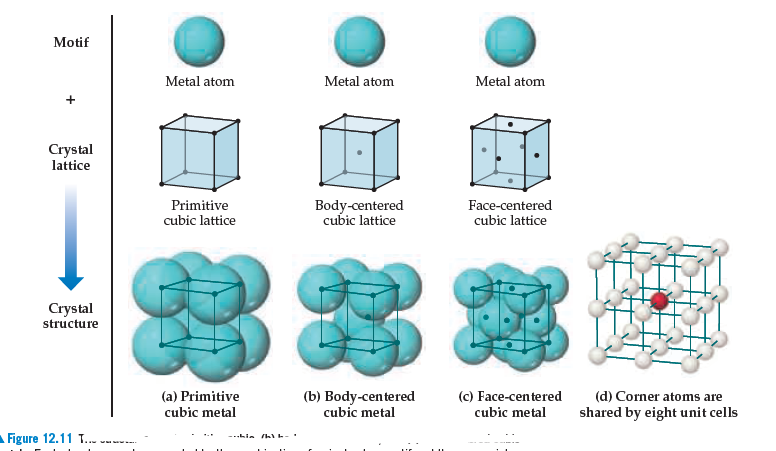
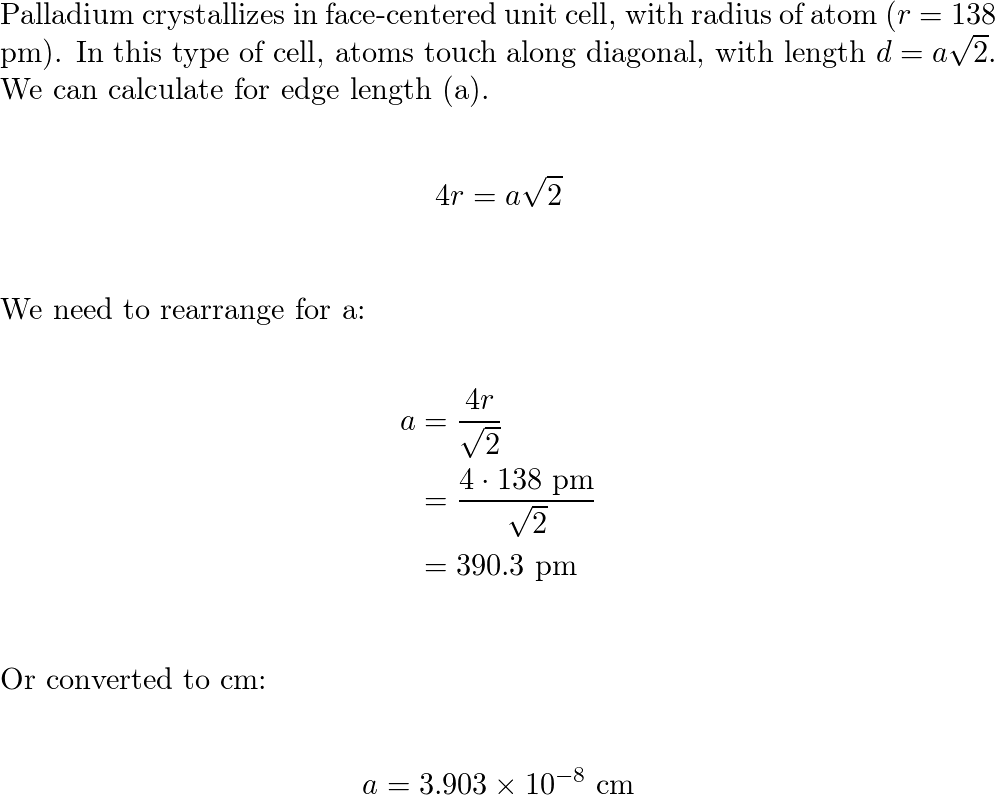Copper crystallizes in a cubic structure. If the density of the metal is 8.% g/cm^3 and the length of the unit cell edge is 361 picometers, find the number of atoms in

A metal crystallizes in the face-centered cubic unit cell with an edge length of 320 pm. \\ A. What is the radius of the metal atom? B. The density of the metal

SOLVED: Palladium crystallizes in face-centered cubic unit cell: Its density is 12.0 gem' at 27*C. Calculate the atomic radius of Pd Solution: Vanadium crystallizes in a body-centered cubic lattice; and the length

OneClass: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is ...

quiz2answers - 1. A metal crystallizes in a face-centered cubic lattice. The radius of the atom is 128 pm and the density of the element is 8.92 g/cm3. | Course Hero

SOLVED: 1. Palladium (at. wt. = 106) crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3. Calculate the atomic radius of palladium and its packing efficiency.

Metallic lead crystallizes in a face-centred cubic lattice, with one Pb atom per lattice point. If the metallic radius of Pb is 175 pm, what is the volume of the unit cell

Face-centered cubic Questions and Answers.pdf - Face-centered cubic problems Problem #1: Palladium crystallizes in a face-centered cubic unit cell. Its | Course Hero
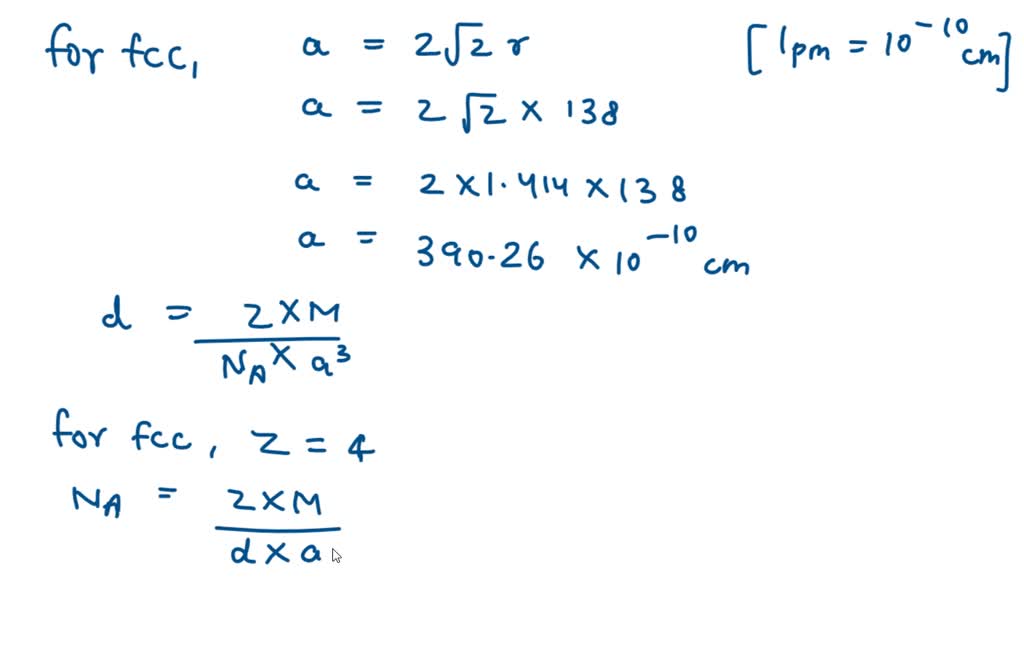
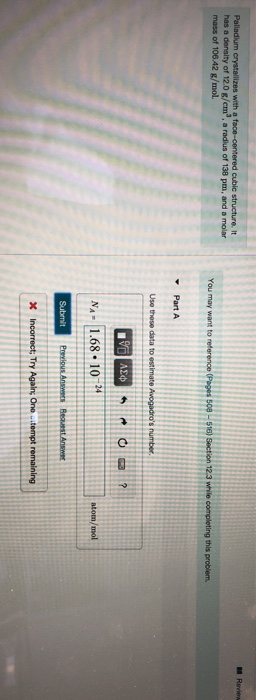
SOLVED: Palladium crystallizes with a face-centered cubic structure. It has a density of 12.0 g / cm^3, a radius of 138 pm, and a molar mass of 106.42 g / mol. Use