FDA warns eye drops may cause infection. Here's a list of 27 products to which the alert applies. - CBS News

Teva Pharmaceuticals USA, Inc., Issues Voluntary Nationwide Recall of Specific Lots of Fentanyl Buccal Tablets CII Because of Labeling Error | ONS Voice

Teva Pharmaceutical | $TEVA Stock | Shares Rally After Winning FDA Approval for Migraine Therapy - Warrior Trading News
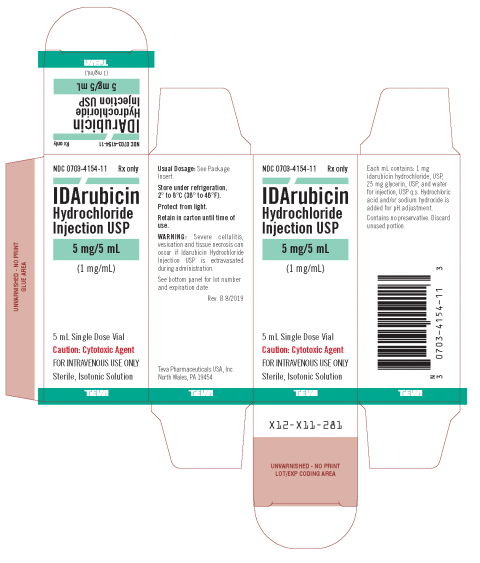
Teva Issues Voluntary Nationwide Recall of One Lot of IDArubicin Hydrochloride Injection USP 5 mg/5 mL Due to the Presence of Particulate Matter | FDA