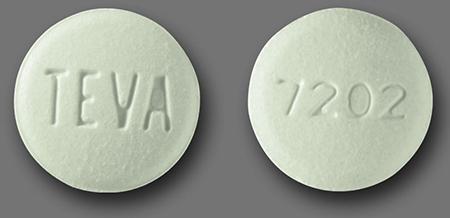
Teva Pharmaceuticals USA recalls Losartan Potassium 25mg and 100mg Tablets USP sold exclusively to Golden State Medical Supply - Pharmaceutical Business review

Drugs recalled after erectile dysfunction medication mixed with antidepressant in packaging 'mix-up' | NewsNation

Teva's recall of U.S.-made drugs latest example of contamination fears in generic marketplace; report says Lilly, Pfizer and former Mylan plant in Morgantown have been cited in the past | WV News

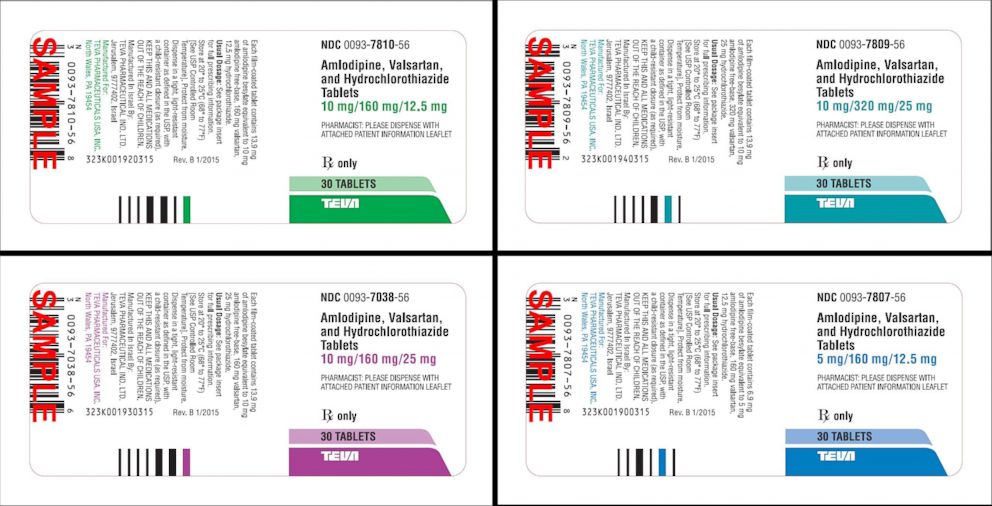
Teva Pharmaceutical Industries: Teva to recall certain blood pressure medicine in U.S., ET HealthWorld

Drugs recalled after erectile dysfunction medication mixed with antidepressant in packaging 'mix-up' | NewsNation
USFDA: Teva Pharmaceutical recalls 38,858 bottles of drugs made by Emcure Pharmaceuticals in US market - The Economic Times
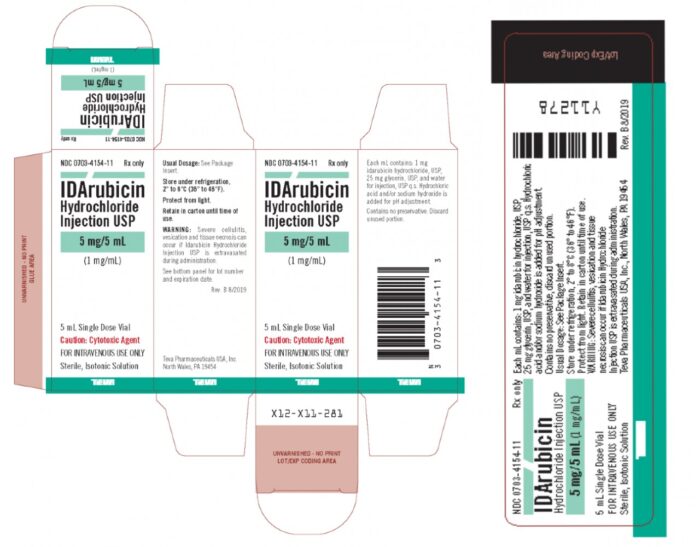
Teva Pharmaceuticals Recalls Acute Myeloid Leukemia Drug Over Particulate Matter Contamination - Top Class Actions

Teva Mold Contamination Problems Result in Recall for 2.5M Drug Vials, Shut Down of California Plant - AboutLawsuits.com